Nuclear envelope fragments meaning1/8/2024 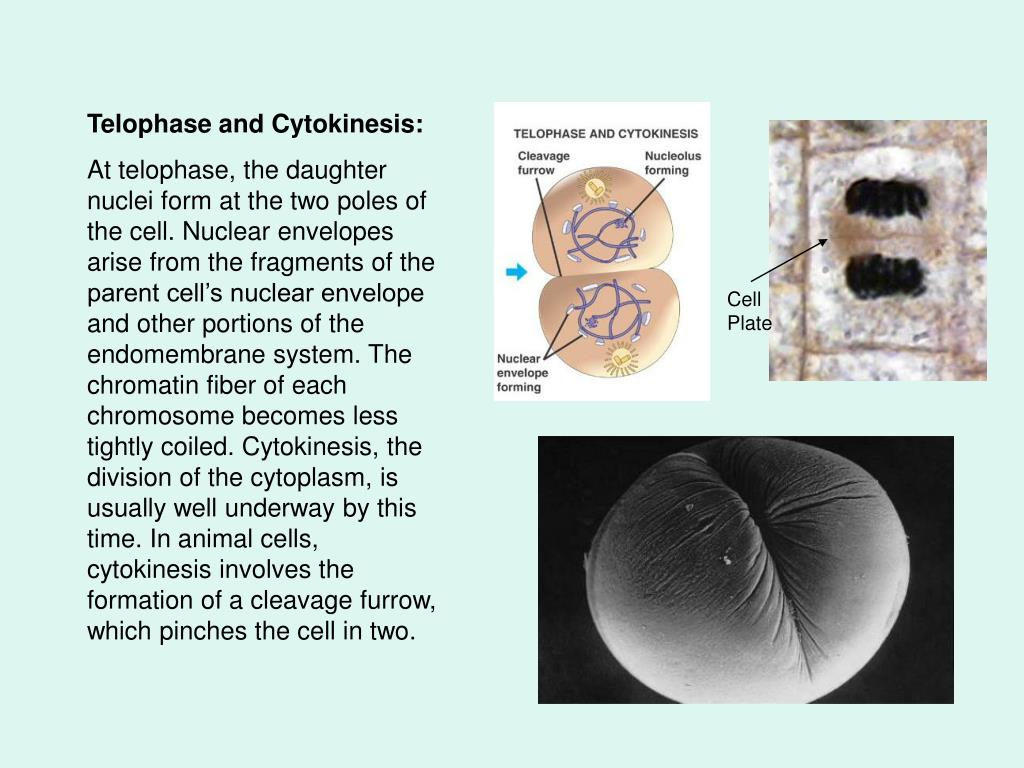
In order to understand how this variability could arise, we have focused on anaphase cells containing lagging chromosomes or chromatin bridges, the most common cause for MN formation. Indeed, Lamin A and NPC seem to have different loading times. ), the presence or absence of Lamin A and other nuclear membrane (NM) components suggested that the loading of Lamin A could be uncoupled from that of nucleoporins. Although the absence of Lamin B1 could indicate that the MN have already been disrupted (as reported by Zhang et al. While Lamin B1 was absent in all MN analysed in both cell lines (as previously reported ), we noticed heterogeneity among the MN in relation to the presence/absence of Lamin A and Importin β or the NPC marker mAb414, which recognizes several FG nucleoporins, with the majority of the MN lacking Importin β and mAb414 (Supplementary Figure 1A, 1B). In order to understand the causes of MN instability, we have analysed the composition of the NE of MN in HeLa and U2OS cells by immunofluorescence. This study proposes a sequential model for the NER where Lamin A targeting to the chromatin is under the control of decreasing cdc2/cyclinB activity (a clock-based model) while NPC loading is spatially regulated by PLK1 activity. Here we show for the first time that, in vertebrates, Polo-like kinase 1 (PLK1) is also participating in the NER control pathway, and that it is essential both for timely disassembly of the NPC at the onset of mitosis and for preventing the loading of the NPC on the chromatin of lagging chromosomes during mitotic exit. Recent studies in Drosophila have shown that an Aurora B gradient appears to regulate a surveillance mechanism that prevents both chromosome decondensation and NER until effective separation of sister chromatids is achieved and to delay Lamin A re-assembly. However, the molecular effectors are still not known. This could be achieved by activation of different phosphatases at different times during mitotic exit (clock model), by the position of the segregating chromatin within the anaphase cell (spatial model) or a combination of both. This ordered assembly during NER suggests that sequential dephosphorylation steps must occur to allow macromolecular complexes to form and target them to the correct place at the right time. During mitotic exit, NE reformation (NER) occurs as a sequential and multi-step process where NUPs assemble to the chromatin earlier than Lamin A but the overall mechanism is still poorly understood (recently reviewed in and ).Ī few studies have shown that this mechanism is regulated by the concerted action of protein phosphatase 1 (PP1) and 2A (PP2A). 
Lamin A phosphorylation by cdc2/cyclinB allows lamina disassembly at the beginning of mitosis while multiple kinases appear to contribute to the disassembly of the NPC via phosphorylation of a crucial target, Nup98. Altogether this study reveals the existence of independent regulatory pathways for Lamin A/C and NPC reorganization during mitosis where Lamin A/C targeting to the chromatin is controlled by CDK1 activity (a clock-based model) while the NPC loading is also spatially monitored by PLK1.ĭuring open mitosis the nuclear envelope breaks down at the end of prophase to allow the separation of sister chromatids and starts to re-assemble during anaphase (reviewed in ). We also provide evidence for the requirement of PLK1 activity for the disassembly of NPCs, but not Lamina A/C, at mitotic entry. Here we show that lagging chromatin, although it can efficiently assemble Lamin A/C, always fails to recruit Nuclear Pore Complexes (NPCs) proteins and that Polo-Like Kinase (PLK1) negatively regulates NPC assembly. Despite its importance, the molecular mechanism at the origin of this instability is still not understood. These problems ultimately lead to a spectrum of chromosome rearrangements called chromothripsis, a phenomenon that is a hallmark of several cancers. These structures are a major source of genetic instability caused by DNA repair and replication defects coupled to aberrant Nuclear Envelope (NE). Micronuclei (MN) arise from chromosomes or fragments that fail to be incorporated into the primary nucleus after cell division. 
Received: AugAccepted: OctoPublished: December 23, 2017 Paola Vagnarelli, email: nuclear envelope polo Like Kinase (PLK1) nuclear pore complex (NPC) lamin A micronuclei chromosome *These authors contributed equally to this work Ines J de Castro 1, 2, *, Raquel Sales Gil 1, *, Lorena Ligammari 1, Maria Laura Di Giacinto 1 and Paola Vagnarelli 1ġCollege of Health and Life Science, Research Institute for Environment Health and Society, Brunel University London, UB8 3PH, UKĢCurrent address: Department of Infectious Diseases, Integrative Virology, University Hospital Heidelberg and German Center for Infection Research (DZIF), 69120 Heidelberg, Germany
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
